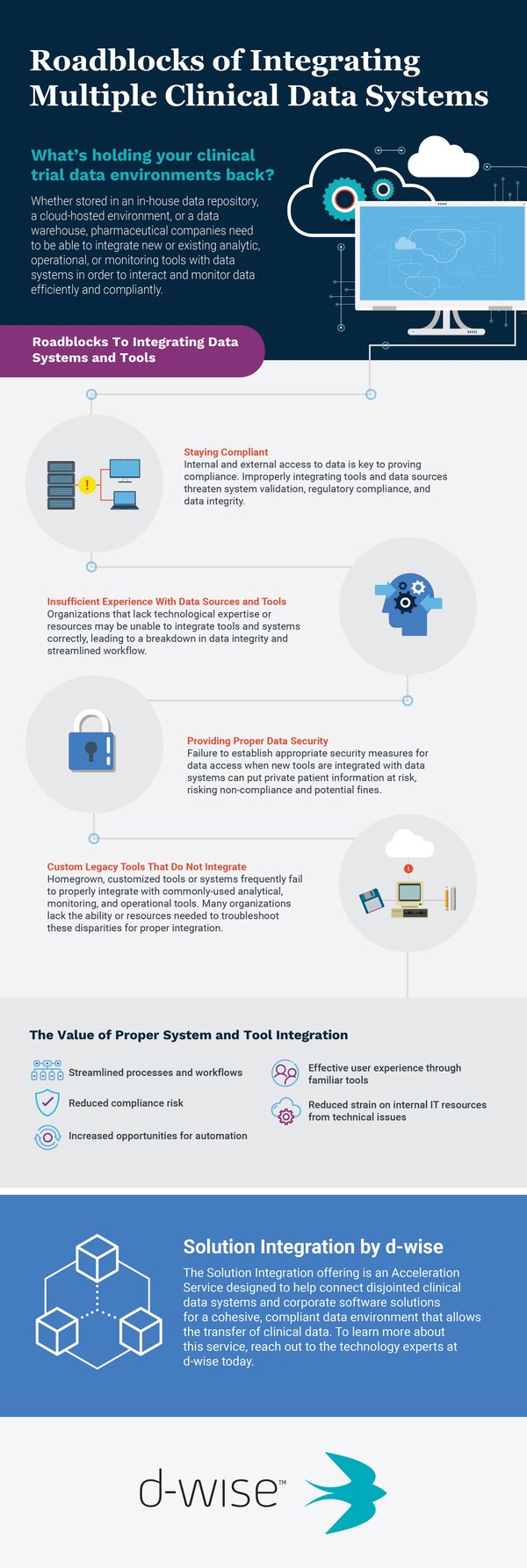
The ability to access and transfer data is one of the key aspects of clinical trial analysis. Whether stored in an in-house data repository, a cloud-hosted environment, or a data warehouse, pharmaceutical companies need to be able to seamlessly interact with data using a wide range of analytical tools.
Discovering if your systems are integrated
There are a number of ways an organization might discover that their data sources, computing system, and tools are improperly integrated. During periods of growth such as mergers, acquisitions, or general organizational success and expansion, many companies may discover the need to integrate a new tool or data source into their current environment. And despite what many inexperienced IT teams may expect, simply dropping a new software into an existing infrastructure is not a viable solution.
Periods of growth are not the only way to discover a lack of system integration. When an organization receives a Form 483 from the FDA, or a letter indicating questionable practices and conditions that do not meet regulatory standards, companies must look inward at their current processes and systems to identify the issues in order to resolve them. In many cases, these processes may be affected by improperly integrated tools that result in non-compliance.
Effects of improperly integrated tools and systems
When software or tools used during analysis are improperly joined with existing data sources, organizations may experience a number of side effects that can cause harm to data integrity and clinical trial timelines. Some effects include:
- Loss of regulatory compliance
- Bottlenecked processes and workflows
- Poor user experience
- Increased strain on IT resources
- Inaccessible data flow
Failure to properly integrate new solutions with existing technologies can have a drastic effect on data integrity. In order to remain confident in the quality of data and its analysis, organizations must ensure that their tools and systems are properly assimilated and work together seamlessly.
Roadblocks to proper system and tool integration
When a new data system or analysis tool is introduced to a data environment, there are several potential roadblocks that may appear that prevent proper integration and implementation. Such roadblocks include:
- An increase of regulations surrounding clinical trial data technologies
- Lack of integration experience relating to modern software, tools, or data sources
- An inability to provide required security on data access
- The use of custom, legacy tools and softwares that do not support full integration
While many organizations may have the ability to handle integration in-house, the amount of time and resources needed to complete proper system integration may not be worth handling on your own. Utilizing the experience and industry expertise of a third-party service provider can save your organization time and effort while simultaneously providing confidence in the integrity of your data.
Solution integration with d-wise
The Solution Integration offering from d-wise was designed to help connect disjointed clinical data systems and corporate software solutions for a cohesive, compliant data environment that allows efficient, effective transfer of clinical data. Backed by industry experts with experience in a wide variety of technologies and systems, this offering can help organizations struggling to improve their environments by handling the heavy lifting, saving you time and resources.
To learn more about this service, reach out to the technology experts at d-wise today.